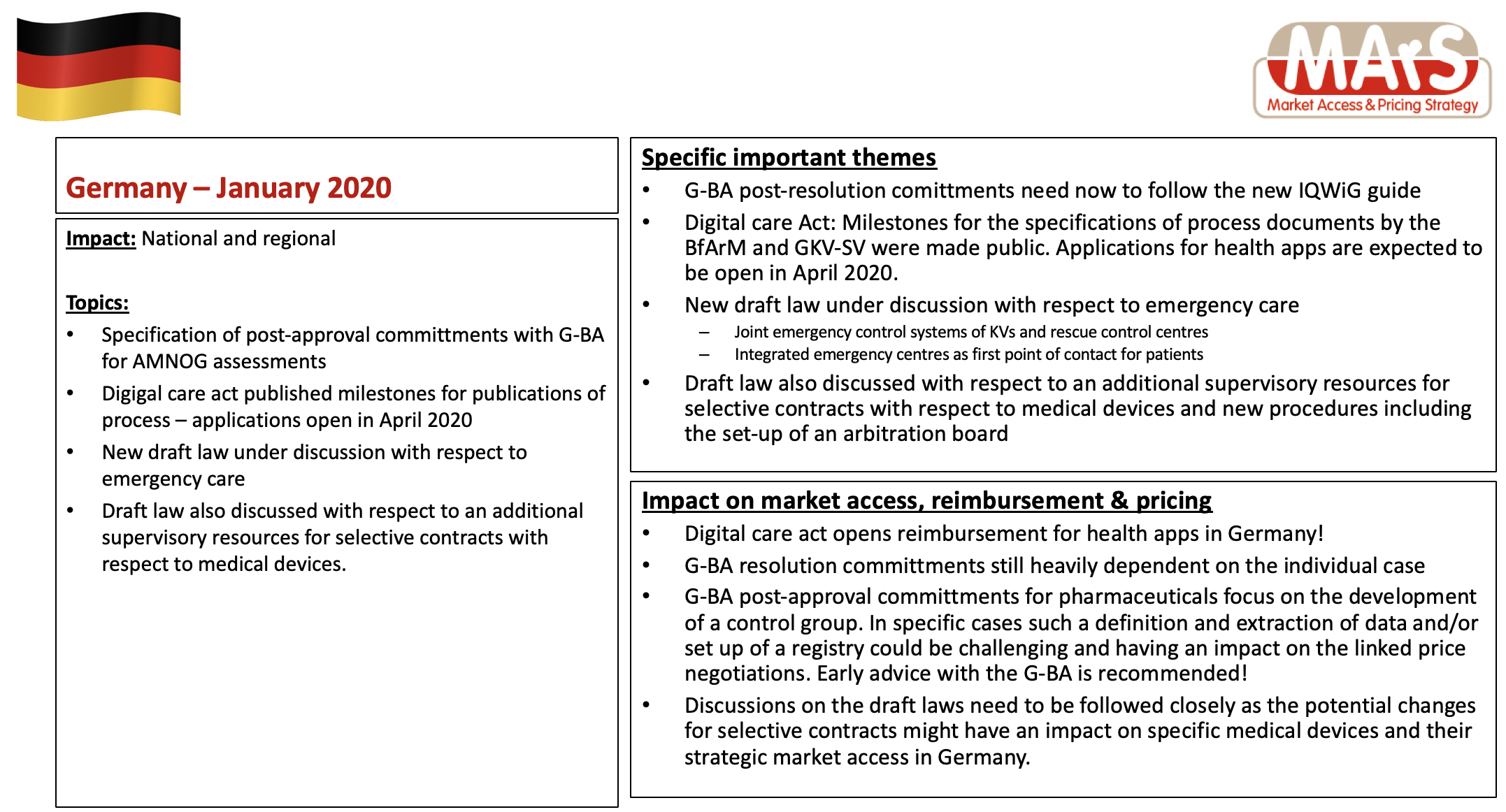
MArS has created another country profile update for Germany as of January 2020 – a summary is available below – the full report is available on request.
As summary:
- G-BA post-resolution comittments need now to follow the new IQWiG guide
- Digital care Act: Milestones for the specifications of process documents by the BfArM and GKV-SV were made public. Applications for health apps are expected to be open in April 2020.
- New draft law under discussion with respect to emergency care
- Draft law also discussed with respect to an additional supervisory resources for selective contracts with respect to medical devices and new procedures including the set-up of an arbitration board
MArS has vast experience in submissions, pricing and negotiations in Germany for pharmaceuticals, medical devices and diagnostics.